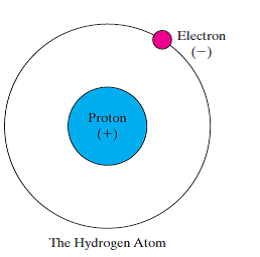
I think we have an old answer on that topic. Some of those other kinds of charge cannot be expressed as simple positive or negative numbers at all, but require more complicated mathematical representation. atoms See all videos for this article More than 90 types of atoms exist in nature, and each kind of atom forms a different chemical element. In 2010, a highly precise measurement of the proton. The proton’s positive charge is equal and opposite to the negative charge on an electron, meaning a neutral atom has an equal number of protons and electrons. (I guess if it didn’t, there wouldn’t be interactions, and we wouldn’t exist if the elementary building blocks of matter didn’t interact with each other). The proton charge radius has been measured since the 1950s using elastic electronproton scattering and ordinary hydrogen atomic spectroscopy. These are a lot like photons (which interact with anything with electrical charge), except that the gluons themselves carry strong-force charge and therefore interact with each other, making a gluey, sticky mess. The strong nuclear force (for which the 2004 Physics Nobel Prize was awarded to Gross, Politzer and Wilczek just a couple of days before I am writing this), involves additional kinds of charge, which interact with the carriers of the strong nuclear force, called gluons. 
The weak nuclear force introduces other kinds of charge (often called "hypercharge" or just weak charge) which are actually related in a very wonderful way to the electrical charges the weak nuclear force and the electromagnetic force are described together in a beautiful model which describes how at very high energies they are one and the same kind of interaction. Other kinds of charge exist! The electromagnetic force is just one of several that particles interact with in nature. We’ve lived with his convention ever since. Electron charge is usually denoted by the symbol e. He based his convention on which signs of charges were deposited by either glass or amber on an object that was rubbed on them. Elastic electron-proton scattering (e-p) and the spectroscopy of hydrogen atoms are the two methods traditionally used to determine the proton charge radius. Electrons have an electric charge of 1, and their mass is approximately about 1/2000 the mass of a neutron or proton. He proposed that electric charge was some kind of fluid, and that objects that gained fluid have positive charge and object that lost it gained a negative charge. The positive charge of the protons is equal to the negative charge of the electrons. In fact, it was Benjamin Franklin who proposed in the mid 1700’s that charge is conserved - that total charge remains the same even though positive charges can be separated from negative ones. The convention of which sign of charge is positive and which is negative is purely arbitrary.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
